Epitomee builds its first production facility
After years of scientific research and development of our simple and safe, game-changing weight loss capsule, it is finally time to transition to mass production and really start to change the way weight loss and metabolic disorders are treated worldwide.
In fact, Nestlé Health Science (NHSc) has decided to partner with us and commercialize our product globally. NHSc’s global reach will bring new exciting opportunities and we are now working hard to build the necessary infrastructure for mass production and adoption of our capsule.
Epitomee’s unique technology
Epitomee’s weight management capsule is safe, non-invasive and very easy to use. After being swallowed it reaches the stomach and shapeshifts into its active configuration; When exposed to the stomach’s pH level the outer shell will dissolve and allow an inner hydrogel scaffold to absorb fluids and expand until it comes into contact with the stomach walls, creating a sense of fullness. Our unique hydro-gel technology can be programmed for a specific duration of operation, after which it dissolves and clears out of the digestive system without leaving a trace.
Quality mass production
Now that development work is done and our device is ready for use, it’s time to focus on establishing a mass production facility to support the massive demand our partnership with Nestlé is going to generate.
As our unique technology and special material characteristics and processes require unique production tools, it was obvious to us that we should develop and build most of the production tools from scratch. To do that we gathered an amazing, dedicated team to build a top-notch facility that will maintain high quality control and quality assurance standards on a mass production scale.
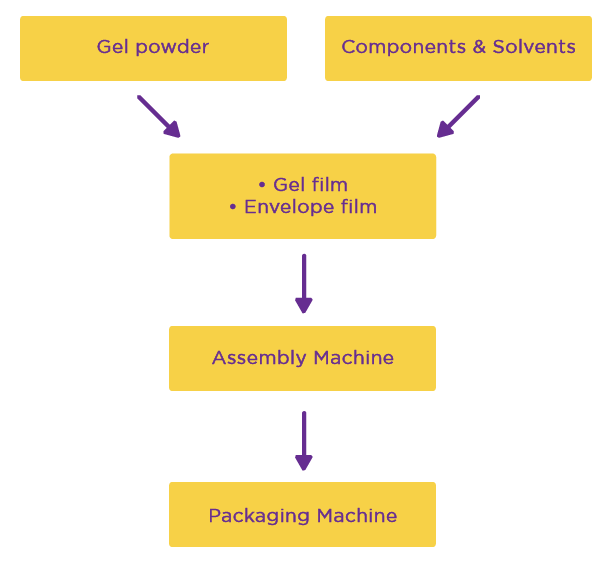
The result is a three-step production line:
Step 1- Making the gel film and envelope film-Epitomee’s proprietary process.
Step 2- Assembling the components into the final product-With the new production systems, combining the two films to make a functional device and inserting it into the capsule can be achieved at a rapid pace.
Step 3: Final packaging- We will be using a fully automated packaging machine to fit our specific needs.
One million dollars were invested so far in designing the production line, assembling it from parts and modules from around the world, and leasing a new space to accommodate it. We traveled internationally to plan, test, verify, and control the mission, despite travel restrictions, to ensure that it was completed on time and according to the required quality.
Step one of the production process will be operational by mid 2022.
Towards the future of ingestible therapeutics
For us this is only the beginning. The core components of our weight management capsule are a part of a platform technology that enable us to develop further products for various applications. We are currently working on a new shape shifting device that can deliver biologic drugs directly to the gut wall, which is attracting a great deal of attention in the medical field: Targeted delivery of biologics.
As part of our goal to contribute to public health and people’s quality of life, we are excited to expand our reach to new indications through new devices.
We’re currently open to partner with innovative companies on enabling our biologics delivery platform to effectively carry their API and optimize its administration for millions of people.

